Helicobacter pylori
Helicobacter pylori is a Gram-negative bacterium that chronically infects the stomachs of over half of the world’s population. It is the strongest known risk factor for developing gastroduodenal ulcers and gastric cancer. To survive in the harsh environment in the stomach, H. pylori has evolved to avoid the microbicidal acid by colonizing a niche close to the epithelial surface. Here, the bacteria are found as two major populations: One is free-swimming and motile within the mucus gel overlying the epithelium, whereas the other is adhered to the surface of epithelial cells and preferentially interacts with the epithelial junctions. The adhered population is associated with worse inflammation and can deliver and inject bacterial virulence factors into epithelial cells to alter host cell biology.
Using polarized cells in culture, we discovered that H. pylori use virulence factors to modify the cell-cell junctions and locally alter cell polarity. This allows the bacteria to grow directly on the cell surface as adherent microcolonies and to persist in conditions that are lethal to free-swimming bacteria. This suggests that cell-associated H. pylori represent a specialized reservoir of bacteria in a niche that is different from the free-swimming population in the mucus. Thus, understanding where and how H. pylori colonizes the gastric epithelium in vivo will yield novel insights on pathogenesis and potential novel targets for therapy.

H. pylori uses the cell surface as a replicative niche.
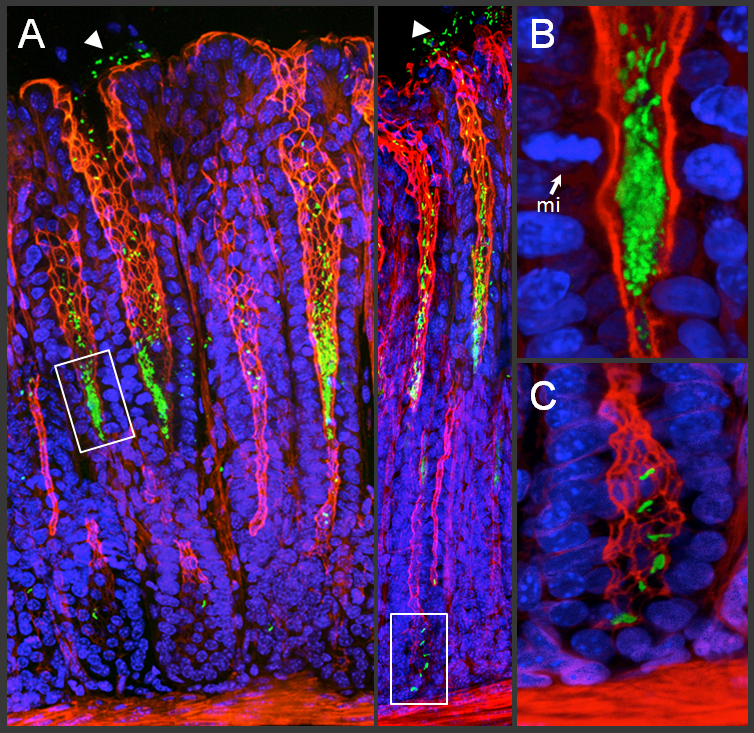
(A) 3D confocal reconstructions of murine gastric mucosa stained to visualize H. pylori (green), host actin (red), and nuclei (blue). H. pylori can be found as free-swimming bacteria in the surface mucus (arrowheads) or attached to the surface of the epithelium in the progenitor and stem cell compartments of the gastric glands (boxes). Gland-associated microcolonies are magnified in (B) in the proliferative zone next to a mitotic (mi) cell, and in (C) in the gland base, where gastric stem cells reside. Images adapted from Sigal et al. 2015, Gastroenterology.
Using an in vivo murine model of H. pylori infection, we discovered that H. pylori grow as distinct bacterial microcolonies deep in the gastric glands and interact directly with gastric progenitor and stem cells. This gland-associated bacterial population is also present in infected human stomachs. H. pylori infection of reporter mice with fluorescently-labeled stem cells revealed that the gland-associated bacteria accelerate gastric stem cell proliferation. This alters epithelial turnover kinetics and leads to glandular hyperplasia. These results demonstrate that direct colonization of the epithelium by H. pylori manipulates stem cell biology and may contribute to the pathogenesis of gastric cancer.
We are interested in further exploring how H. pylori is able to colonize this specialized niche in the gastric glands and how this affects host cell biology.
How do H. pylori detect and find the epithelium in the gastric glands?
Does gland colonization confer benefits to the bacteria that are not available in the surface mucus?
What bacterial factors are required for H. pylori to persist on the epithelium in the gastric glands?
What host and/or environmental factors control H. pylori localization to the gastric glands?
How does H. pylori infection alter the functionality and gene expression of gastric stem cells?
Helicobacter pylori is a Gram-negative bacterium that chronically infects the stomachs of over half of the world’s population. It is the strongest known risk factor for developing gastroduodenal ulcers and gastric cancer. To survive in the harsh environment in the stomach, H. pylori has evolved to avoid the microbicidal acid by colonizing a niche close to the epithelial surface. Here, the bacteria are found as two major populations: One is free-swimming and motile within the mucus gel overlying the epithelium, whereas the other is adhered to the surface of epithelial cells and preferentially interacts with the epithelial junctions. The adhered population is associated with worse inflammation and can deliver and inject bacterial virulence factors into epithelial cells to alter host cell biology.
Using polarized cells in culture, we discovered that H. pylori use virulence factors to modify the cell-cell junctions and locally alter cell polarity. This allows the bacteria to grow directly on the cell surface as adherent microcolonies and to persist in conditions that are lethal to free-swimming bacteria. This suggests that cell-associated H. pylori represent a specialized reservoir of bacteria in a niche that is different from the free-swimming population in the mucus. Thus, understanding where and how H. pylori colonizes the gastric epithelium in vivo will yield novel insights on pathogenesis and potential novel targets for therapy.

H. pylori uses the cell surface as a replicative niche.

(A) 3D confocal reconstructions of murine gastric mucosa stained to visualize H. pylori (green), host actin (red), and nuclei (blue). H. pylori can be found as free-swimming bacteria in the surface mucus (arrowheads) or attached to the surface of the epithelium in the progenitor and stem cell compartments of the gastric glands (boxes). Gland-associated microcolonies are magnified in (B) in the proliferative zone next to a mitotic (mi) cell, and in (C) in the gland base, where gastric stem cells reside. Images adapted from Sigal et al. 2015, Gastroenterology.
Using an in vivo murine model of H. pylori infection, we discovered that H. pylori grow as distinct bacterial microcolonies deep in the gastric glands and interact directly with gastric progenitor and stem cells. This gland-associated bacterial population is also present in infected human stomachs. H. pylori infection of reporter mice with fluorescently-labeled stem cells revealed that the gland-associated bacteria accelerate gastric stem cell proliferation. This alters epithelial turnover kinetics and leads to glandular hyperplasia. These results demonstrate that direct colonization of the epithelium by H. pylori manipulates stem cell biology and may contribute to the pathogenesis of gastric cancer.
We are interested in further exploring how H. pylori is able to colonize this specialized niche in the gastric glands and how this affects host cell biology.
How do H. pylori detect and find the epithelium in the gastric glands?
Does gland colonization confer benefits to the bacteria that are not available in the surface mucus?
What bacterial factors are required for H. pylori to persist on the epithelium in the gastric glands?
What host and/or environmental factors control H. pylori localization to the gastric glands?
How does H. pylori infection alter the functionality and gene expression of gastric stem cells?